What is Palmitoylethanolamide?
Palmitoylethanolamide (PEA) is an endogenous fatty acid amide, a naturally occurring compound that plays a significant role in intracellular signaling mechanisms. In both humans and animals, palmitoylethanolamide is produced primarily as a biological repair mechanism, and thus serves as an effective modulator of inflammation and chronic and/or nerve pain. Among the compound’s numerous beneficial mechanisms are neuroprotective and anti-inflammatory capabilities, as well as lipid modulating actions.
In mid-1950s, researchers examining the therapeutic effects of yolk in passive Arthus phenomenon, or anaphylactic arthritis, identified palmitoylethanolamide as a component in chicken egg yolk. Palmitoylethanolamide was also discovered in soybean lecithin and peanut meal shortly thereafter. Since then, numerous clinical trials have extracted the substance from various biological materials and validated its efficacy as a nutraceutical, or a pharmaceutical nutrient that yields beneficial therapeutic effects and can be found in plant and/or animal sources.
Palmitoylethanolamide’s mechanism of action lies in its ability to bind to peroxisome proliferator-activated receptors in cell nuclei, which allows the compound to perform a series of biological functions related to metabolic functioning, as well as chronic and neuropathic pain and inflammation. To date, more than 350 clinical research papers have elucidated the physiological properties and therapeutic capabilities of palmitoylethanolamide, as well as its efficacy and tolerability.
Palmitoylethanolamide Benefits and Uses
Fibromyalgia
A 2015 study published by Pain Therapy evaluated palmitoylethanolamide’s ability to modulate the symptoms of fibromyalgia, a syndrome characterized by chronic, persistent pain that is often resistant to typical analgesic therapies. This study specifically observed the effects of duloxetine, an anti-depressant, and pregabalin, an anxiolytic and anti-convulsant, alongside palmitoylethanolamide in terms of anti-inflammatory, analgesic, and pain-relieving effects. The project was two-fold; the researchers first executed a retrospective observational study of a patient group receiving duloxetine and pregabalin for six months. The second step involved a prospective study plus PEA administration for three months. The results indicated a decrease in pain evoked and pain intensity after three months in the initial retrospective. In the prospective observational study, duloxetine + pregabalin + palmitoylethanolamide yielded a significant improvement in pain symptoms, with a greater reduction in symptoms in compared to the duloxetine and pregabalin treatment alone. Further, no negative side effects were observed.
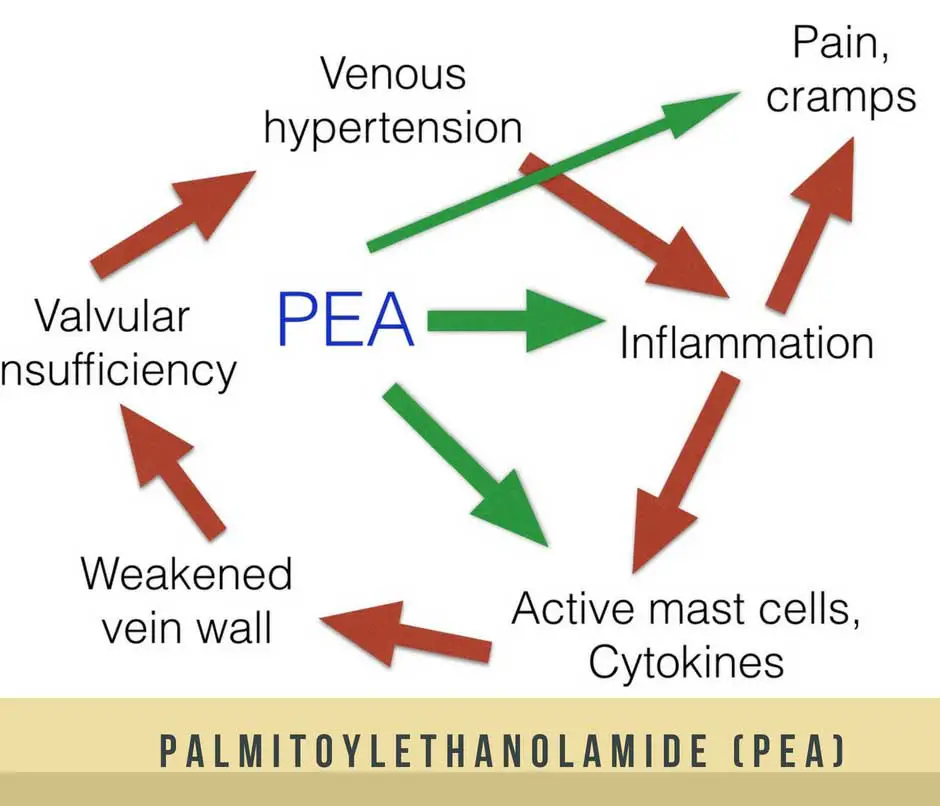
Palmitoylethanolamide’s mechanism of action in pain modulation Is relatively well-understood. Clinical animal models have suggested that palmitoylethanolamide acts on two processes that arouse chronic nerve pain: nerve sprouting and dorsal root ganglia activation. Nerve sprouting refers to the growth of axons from a damaged neuron that projects to denervated muscle areas. Dorsal root ganglia activation involves the stimulation of sensory neurons at the dorsal roots of spinal nerves.
Depression/Anxiety
Some clinical research points to palmitoylethanolamide as a potential adjuvant therapeutic in the treatment of anxiety and/or depression. A 2013 study published by CNS & Neurological Disorders – Drug Targets examined the antidepressant effect of a compound formed from co-ultramicronized palmitoylethanolamide and luteolin, a naturally occurring flavonoid. Laboratory mice exhibiting chronically anxious/depressive behavior were administered six weeks of a palmitoylethanolamide + luteolin treatment, and were then evaluated on parameters of behavior, neurogenesis, neuroplasticity, neurotrophic, and apoptotic protein expression. Results indicated that PEA + luteolin exhibited a significant antidepressant effect at a relatively low dosage of 1 mg/kg.
In a recent 2018 study published by the Journal of Affective Disorders, researchers observed the antidepressant effects of palmitoylethanolamide in laboratory models of chronic depression. In a randomized, placebo-controlled study, researchers administered 600 mg twice daily of palmitoylethanolamide (or placebo) alongside the antidepressant citalopram to 58 depressive patients for six weeks. By week two, the palmitoylethanolamide group demonstrated a significant reduction in symptoms on the Hamilton Depression Rating Scale in comparison to the placebo group, and these improvements continued throughout the trial period. Researchers concluded that palmitoylethanolamide may be effective as an adjunctive therapy to psychiatric antidepressant medications in patients with major depressive disorder.
While palmitoylethanolamide should not be administered as a replacement for psychiatrist-prescribed antidepressant medications, studies such as those described above indicate that it may be an effective additional treatment with rapid-onset therapeutic effects.
Nootropic
Some clinical research has elucidated palmitoylethanolamide’s role as a nootropic, or a cognitive booster facilitating improved information synthesis and retention. A 2018 study published by Translational Psychiatry evaluated the role of ultramicronized palmitoylethanolamide in ameliorating cognitive decline and memory impairment in Alzheimer’s disease. Results of the laboratory mouse model yielded that palmitoylethanolamide normalized astrocytic function, rebalance glutamatergic transmission, and restrained neuro-inflammation, ultimately resulting in improved learning, memory, and immunodefense. Researchers noted that palmitoylethanolamide administration was particularly effective in younger mice, suggesting that it may have potential as an early therapeutic in the treatment of Alzheimer’s dementia.
Similar research has demonstrated that palmitoylethanolamide produces a neuroprotective effect in patients with Alzheimer’s-induced memory and learning impairments. A 2012 study published by Neuropsychopharmacology examined palmitoylethanolamide’s role in modulating Amyloid-β25-35-induced cognitive impairments in a mouse model of Alzheimer’s disease. Results indicated that palmitoylethanolamide reduced lipid peroxidation, protein nystrosylation, inducible nitric oxide synthase induction, and caspase3 activation, ultimately resulting in a “rescue” of memory deficits and behavioral impairments induced by Amyloid-β25-35.
A 2017 in vivo study evaluated palmitoylethanolamide’s ability to regulate cognition, recognition memory, and affective processing in the mesolimbic dopamine system. Using a combination of in vivo electrophysiology and behavioral pharmacological assays in laboratory rats, researchers were able to determine that palmitoylethanolamide produced a hyper-dopaminergic activity state in the mesolimbic system, ultimately impacting social interaction and recognition memory, spatial location, and context-independent associative fear memory formation. Researchers concluded that, by modulating GPR55 receptor signaling, palmitoylethanolamide may yield a potent nootropic effect.
Chronic Regional Pain Syndrome (CRPS)
Chronic regional pain syndrome (CRPS) refers to a condition characterized by persistent, debilitating, and often-untreatable pain, changes to the quality of the afflicted individual’s hair, skin, and nails, tremors/muscle spasms, and insomnia. In rare and severe cases, individuals suffering from CRPS may experience ulcers, muscle atrophy, and/or muscle contractures.
Among new treatments being explored for CRPS is palmitoylethanolamide administration. A 2013 study published by the Journal of Pain Research evaluated the therapeutic efficacy of palmitoylethanolamide and topical ketamine cream in a patient suffering from intractable CRPS type 1. Researchers found that palmitoylethanolamide combined with ketamine 10% cream down-regulated mast cells, resulting in the inhibition of inflammatory instigators such as histamine, TNF-alpha, prostaglandins, and serotonin in vivo. In vitro, researchers observed that the patient exhibited marked reduction in swelling and skin discoloration after one month of treatment, plus a greater ability to walk independently. They concluded that the palmitoylethanolamide and ketamine topical treatment was effective with minimal side effects. However, this represents a study evaluating a single patient, so further research clinical research must be conducted to verify the efficacy of this treatment.
Nerve Pain
Nerve pain encompasses a wide range of chronic symptoms that are induced by damage to the nerves that transfer information from the spinal cord to the skin and muscles. Palmitoylethanolamide is well-understood to be an effective pain modulator, and thus has emerged in fairly recent research as a possible therapeutic agent for several types of chronic neuropathic pain. Palmitoylethanolamide is considered one of the best supplements to naturally deal with nerve pain.
A 2012 study conducted by Pain Medicine evaluated the efficacy of palmitoylethanolamide in treating a sample of 610 patients who were unable to control or ameliorate their chronic pain by standard analgesic therapies. After administering 600 mg of palmitoylethanolamide twice daily for three weeks, researchers observed that the treatment significantly decreased pain intensity in all patients without the aid of any concomitant standard pain therapies.
A 2011 study published by PLOS (Public Library of Science) One yielded similar results. Researchers examined the anti-inflammatory capabilities of palmitoylethanolamide in the human skeletal muscle. Subjects were eleven women with chronic neck and/or shoulder pain. In muscle dialysate samples collected from the trapezius muscles, palmitoylethanolamide was clearly detectable and higher in myalgic subjects in comparison to the healthy control subjects. Researchers concluded that palmitoylethanolamide may represent a foundation from which nociceptice and antinociceptive (referring to pain arising from the stimulation of nerve cells) processes in the muscle can be better understood and managed.
A more recent 2017 study published by the Korean Journal of Anesthesiology evaluated the effects of palmitoylethanolamide on inflammatory and neuropathic pain in laboratory rats. Researchers administered von Frey filaments and wet the plantar surfaces of laboratory rats with acetone in order to measure tactile and cold sensitivity. Results yielded that hyperalgesia, or an increased sensitivity to pain as a result of chronic inflammation, was significantly decreased by both intraperitoneal and intra-articular injections of palmitoylethanolamide.
Glaucoma
Some recent research has pointed to palmitoylethanolamide as a potential natural retinoprotectant, or an agent that can combat pathogenic threats to the retinas and thus the eyesight. A 2015 review published by the Journal of Opthalmology evaluated palmitoylethanolamide’s therapeutic effects in the treatment of glaucoma, the second most prevalent cause of blindness. One randomized clinical trial in the review examined the effects of orally administered palmitoylethanolamide in forty-two patients with elevated intraocular pressure (or IOP, a significant contributing factor in glaucoma development) and found that oral PEA significantly reduced the markers of IOP after two months of treatment.
Another placebo-controlled clinical trial, published in 2013, explored the effects of palmitoylethanolamide in forty patients experiencing ocular hypertension and endothelial dysfunction of the retinas. Researchers concluded that an administration of 600 mg per day of palmitoylethanolamide significantly improved flow-mediated dilation measurements and intraocular pressure measurements in the control group. This study took place over a period of three months and was followed up after a two-month wash-out period.
Arthritis
A 2017 study published by BMC Veterinary Research evaluated the therapeutic effects of palmitoylethanolamide and quercetin, a plant flavonoid, in the treatment of osteoarthritis in animal subjects. Researchers administered an oral composite of palmitoylethanolamide co-ultramicronized with quercetin (PEA-Q) to laboratory rats exhibiting inflammatory and osteoarthritic pain and found that PEA-Q decreased both inflammatory and hyperalgesic responses. Further, PEA-Q improved locomotor function, protected cartilage against histological damage, and reduced mechanical allodynia.
Another study, published in 2013 by Arthritis Research & Therapy, explored the therapeutic potential of palmitoylethanolamide in a laboratory animal model of mice injected with collagen type II. After administering 10 mg/kg of ultramicronized palmitoylethanolamide, researchers found that oxidative and nitrosative damage was significantly decreased in PEA-treated mice. Further, plasma levels of proinflammatory cytokines were heavily reduced after treatment.
Influenza and/or Common Cold
Though further research is needed to produce a well-rounded pharmacological profile of efficacy, some clinical evidence has indicated that palmitoylethanolamide may have an anti-influenza mechanism of action. A research review published by the International Journal of Inflammation evaluated data synthesized from PubMed over the past fifty years and found six clinical trials focusing on palmitoylethanolamide’s therapeutic efficacy in the treatment of respiratory inflammation and flu. Available evidence points to palmitoylethanolamide as an entity capable of attenuating intestinal injury and inflammation and inhibiting proinflammatory cytokine production in virulent viruses. Researchers suspected that PEA may be able to de-escalate influenza and cold symptoms by down-modulating inflammatory cytokines and thus restraining disease progression.
Palmitoylethanolamide Dosage
In a 2017 safety evaluation, palmitoylethanolamide was determined not to induce genotoxic effects in human cells at doses of 1,000 mg/kg for a preliminary 14-day study. The LD50, or lethal dose, was greater than 2,000 mg/kg. Palmitoylethanolamide is typically available in capsule form, often at a standard dosage of 400 mg per pill, taken twice or thrice daily. However, it is best to consult your doctor for this.
Palmitoylethanolamide can be ingested in powder and/or capsule form. It is also available as a non-steroidal topical cream for the purposes of alleviating pain and inflammation.
Palmitoylethanolamide Side Effects, Safety, Dangers and Warnings
To date, no serious side effects or drug-to-drug interactions have been reported as a result of palmitoylethanolamide supplementation. Individuals who are pregnant or nursing, have experienced severe hepatic or renal failure, or are under the age of eighteen should consult a physician thoroughly before beginning a palmitoylethanolamide regimen due to a lack of research regarding its safety in these instances.
Overall palmitoylethanolamide has a very limited side effects profile. Some individuals have reported gastrointestinal discomfort or a feeling of “heaviness” in the stomach after consumption.
It is important to note that palmitoylethanolamide tablets that are not labeled as “micronized” or “ultramicronized” are associated with reduced therapeutic efficacy. Such products may be impure and potentially unfit or even dangerous for consumption.
What Natural Foods Contain Palmitoylethanolamide (PEA)?
As a fat-derived compound, palmitoylethanolamide (PEA) [1] is found in a wide range of food products.[2] You can find PEA in almost every food you consume daily, particularly those with high fatty-acid contents, from egg yolks to peanuts. However, you can find them in large amounts in the following produce:
- Soy lecithin
- Soybean
- Roasted coffee
- Alfalfa
- Wheat flour
- Walnuts
- Pearl barley
- Peanuts
- Black-eyed peas
- Brocolli
If you’re planning to bulk up on PEAs, consider incorporating these food items into your daily diet. However, you must keep in mind to consume this product within the recommended levels. Exceeding the dietary regulations could result in side effects.
Since PEAs are fat-based compounds, it’s unusual to find them in carbohydrate and fiber-based products. Regardless, they are still present but in relatively minute amounts. The list below shows food sources containing small amounts of PEA.
- Bovine milk
- Elk milk
- Potatoes
- Lentils
- Apple
- Orange
- Grapes
- Almonds
A PEA-rich diet is best complemented with regular exercise and a balanced lifestyle. You can also take PEA supplements to ensure you’re getting this essential nutrient correctly. When taken together, these three factors will help improve your overall health and well-being.
Frequently Asked Questions (FAQ)
Why are both Palmitoylethanolamide and Phenethylamine Called PEA?
Palmitoylethanolamide and Phenethylamine are completely different supplements and unfortunately hold the same abbreviation of PEA. Even Wikipedia calls both supplements PEA. Palmitoylethanolamide is an endogenous fatty acid while Phenethylamine is a natural monoamine alkaloid.
How To Take Palmitoylethanolamide Powder?
How you take your PEA powder varies per manufacturer. It’s best to follow the directions stated on the product label. However, the general direction recommends users dissolve and ingest 400mg of PEA powder daily.
What Is Ultramicronized Palmitoylethanolamide?
Ultramicronized palmitoylethanolamide (UPEA)[3] is a type of PEA with smaller particle sizes. The smaller particle size allows for quicker absorption and higher bioavailability in the body.
Does Palmitoylethanolamide Cause Constipation?
PEA may cause constipation[4] in some individuals. If you experience this side effect, increase your water intake and add more fiber-rich foods to your diet. You can also speak with your physician for expert advice.
Can Palmitoylethanolamide Cause Headaches?
Some people report experiencing headaches when taking PEA supplements. If you experience this side effect, drink plenty of fluids and get some rest. It’s also best to consult your doctor to rule out any other potential causes.
Can Palmitoylethanolamide Cause Weight Gain?
PEA is a fat-based compound, so it’s possible that it could cause weight gain in some individuals. However, this side effect is rare, and the weight gain[5] is usually minimal. If you are concerned about this, speak with your doctor.
Does Palmitoylethanolamide Make You Sleepy?
PEA does not generally cause drowsiness or sleepiness. However, if you experience this side effect, it’s best to consult your doctor. Side effects may vary per individual.
Can Palmitoylethanolamide Be Used for Long COVID?
There is currently no evidence to support using PEA for long COVID. However, PEA effectively treats other conditions, such as pain, inflammation, and anxiety. If you are experiencing symptoms of long COVID, it’s best to consult your doctor.
Can Palmitoylethanolamide Be Given to Dogs and Cats?
Yes, PEA can be given to dogs and cats[6]. However, it’s best to consult your veterinarian before giving your pet any supplements for the appropriate dosing.
References:
[1] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8157570/
[4] https://journals.sagepub.com/doi/10.1177/1721727X15575869
[5] https://pubmed.ncbi.nlm.nih.gov/24428531/
[6] https://www.mdpi.com/2076-2615/11/4/952
https://www.ncbi.nlm.nih.gov/pubmed/13211905
https://www.ncbi.nlm.nih.gov/pubmed/23844686
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3358748/
https://www.ncbi.nlm.nih.gov/pubmed/26334329
http://journals.plos.org/plosone/article/citation?id=10.1371/journal.pone.0027257
https://www.ncbi.nlm.nih.gov/pubmed/29486338
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3744360/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3500919/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3500919/
https://www.ncbi.nlm.nih.gov/pubmed/24246048
https://www.hindawi.com/journals/iji/2013/151028/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4667059/#sec2title
https://www.ncbi.nlm.nih.gov/pubmed/28861501
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4336979/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5332261/
https://bpspubs.onlinelibrary.wiley.com/doi/pdf/10.1111/bph.13580
https://clinicaltrials.gov/ct2/show/NCT01491191




thanks for an amazing article!