Arachidonic Acid (AA) is a 20-carbon chain polyunsaturated fatty acid with four methylene-interrupted cis double bonds. It is an integral constituent of the biological cell membrane, and it is necessary for the function of all cells. AA is particularly abundant in the brains, muscles, and livers of organisms. It can be obtained from food (such as poultry, animal organs, and meat, fish, seafood, and eggs), or by desaturation and chain elongation of the essential fatty acid, linoleic acid [11].
Due to its inflammatory potential in the muscles, AA is most commonly sold as a supplement for bodybuilders to increase lean muscle gain. Worth noting here is that inflammation in the muscles can be a good thing for athletes; it is a major regulator of growth and promotes training adaptations such as satellite cell proliferation, which contributes to fuller musculature [12]. Several studies have indicated AA’s efficacy in muscular proliferation and reparation of muscle tissue, but its safety as a long-term supplement is poorly understood and requires more clinical evaluation [13].
With this being said, AA exhibits a myriad of other essential functions, and it is pivotal for the efficacious functioning of the brain, liver, and many other organs. Because of its role in inflammation, its metabolic pathway is often targeted in the treatment of arthritis, allergies, and asthma. Some clinical studies also indicate that AA may be useful in the treatment of conditions as diverse as alopecia and chronic long-term depression1.
Arachidonic Acid Function
Cell membrane fluidity
Arachidonic Acid’s four methylene-interrupted cis double bonds endow it with high mobility and allow the substance flexibility and selective permeability to membranes. In particular, AA’s fluidity influences the function of numerous membrane proteins involved in cellular signaling. Further, AA plays a fundamental role in the maintenance of cell integrity and vascular permeability. Studies indicate that these factors and activities may explain AA’s key roles in terms of neuron function and brain synaptic plasticity1.
Receptors and enzymes
AA enhances the functional activity of ligand-gated ion channels (LGICs) – specifically the γ-amino butyric acid receptor (GABA-R) – located on the neural membranes of cells. LGICs are essential proteins that regulate the flow of selected ions across plasma membranes. AA is further implicated in the activation of eosinophils (disease-fighting white blood cells), neutrophils (the most abundant type of white blood cell in humans), and macrophages (phagocytes that specialize in the removal of cellular debris like dead cells)1.
Cell death
As discussed, polyunsaturated fatty acids – especially Arachidonic Acid – affect the function of numerous ion channels, the activities of several different enzymes, and are implicated in cell apoptosis, necrosis, and death. While the cytotoxicity of Arachidonic Acid is, as Pompeia et al. put it, “undeniable,” it is also one of its most crucial and beneficial functions in vivo. For example, cell apoptosis and death are of critical importance during embryogenesis, meaning that the activities of AA are highly impactful on the health of newborns (relevantly, AA is also an important component of human milk)1.
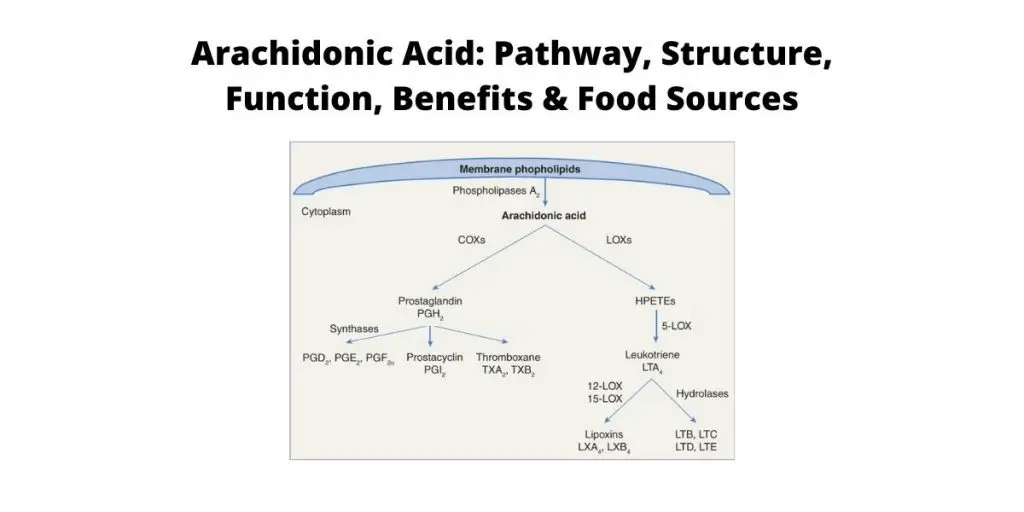
Arachidonic Acid Metabolism Pathway
The Arachidonic Acid pathway plays a pivotal role in cardiovascular biology, carcinogenesis, and inflammatory diseases such as asthma and arthritis1. In scientific terms, here is how it works: esterified (this refers to a chemical event in which two reactants form an ester) AA on the inner surface of the cell membrane is hydrolyzed by phospholipase A2 (PLA2), which is then further metabolized by cyclooxygenases (COXs), lipoxygenases (LOXs), and cytochrome P450 (CYP) enzymes. These reactions produce a wide variety of bioactive mediators that includes prostanoids, leukotrienes (LTs), dihydroxyeicosatetraenoic acid (diHETEs), and lipoxins (LXs). Many of these entities have demonstrated promise as therapeutic targets for cardiovascular diseases, cancers, and inflammatory conditions [14].
As previously mentioned, COXs are the first enzymes to metabolize AA. They require the release of AA from a cell’s plasma membrane by phospholipases and induce the subsequent metabolization of AA by PGG2 and PGH2 (two COX enzymes). There are two primary COX isoforms (referring to two or more functionally similar proteins): COX-1, which is expressed in most cells and serves as the dominant source of prostanoids (which are mediators of inflammatory and anaphylactic reactions), and COX-2 (or PGTS2), which is a more potent source of prostanoid formation in proliferative diseases like cancer4.
The LOX metabolic pathway follows COX metabolism. In this process, Arachidonate 5-LOX (ALOX5) and LT receptor antagonists (or antileukotrienes) proliferate and have been harnessed and refined for the treatment of asthma and seasonal allergies4.
The third metabolic route in AA metabolism is the cytochrome P450 (CYP) pathway. This process exhibits an array of hyprolase and epoxygenase functions and thus produces a wide mixture of products. For example, the ω-hydroxylase activity of CYP enzymes converts AA to hydroxyeicosatetraenoic acids (HETEs), some of which have been demonstrated to possess pro-inflammatory effects in addition to contributing to vascular function. CYP-derived epoxyeicosatrienoic acids (EETs) also regulate cell proliferation, angiogenesis, and metastasis, in addition to influencing capillary formation in endothelial cells. As a result, CYP-derived Arachidonic Acid metabolites have the ability to regulate tumor growth, progression, and metastasis4.
Arachidonic Acid Food Sources
Arachidonic acid is obtained from food sources such as poultry and other animal meats, fish, seafood, and eggs. However, AA is also accessible and essential to herbivores and vegetarians, as it is a central component of linoleic acid, an essential fatty acid abundant in many nuts, seeds, and vegetable oils1.
Benefits of AA
Muscle Growth and Bodybuilding
In particular, Arachidonic Acid has been reputed to impact body composition, muscle function, and power output in strength-training individuals [15]. This is in part because AA makes up between 15-17% of total fatty acids in skeletal muscles1.
In an investigation published by the Journal of the International Society of Sports Nutrition, researchers examined the effects of AA supplementation on performance and body composition in strength-trained males. Additionally, they performed a secondary study looking into the molecular responses of AA supplementation following acute exercise in laboratory rats. In the first part of the study, thirty strength-trained males were divided into AA and placebo groups and underwent an eight-week, three-day per week, non-periodized training protocol. Researchers assessed quadriceps muscle thickness, whole-body composition, and general muscle strength at baseline and post-exercise test. In the rodent model, male Wistar rats were divided into AA or water groups for eight days and were then strength trained via electrical stimulation on unilateral plantar flexions. In both instances, findings indicated that AA supplementation positively augmented strength-training induced adaptations in human males and male Wistar rats [5].
In another study conducted by the Journal of Applied Physiology, nineteen resistance-trained young men (all of whom had less than a year of experience with lower body resistance training) were randomized to consume 1.5 g/day of AA or a placebo for four weeks. Researchers found that four weeks of AA supplementation did not alter the early myofibrillar MPS response following acute exercise. However, following approximately forty-eight hours of recovery, markers of ribosome biogenesis were increased in men receiving AA supplementation compared with placebo. Similarly, the percentage change in muscle satellite cell number tended to be higher in the AA group than with placebo. Researchers concluded that AA supplementation did not appear to modulate early anabolic responses to exercise, but it did modulate the degree of tissue remodeling during the later stages of exercise recovery 6.
Read Also: Beta-Ecdysterone: Bodybuilding’s Secret Supplement Reviewed.
Inflammation
As was previously discussed, AA is mainly metabolized by three enzymes: COX, LOX, and CYP450. Based on these metabolic pathways, AA can be converted into various metabolites that trigger different inflammatory responses 4. Many studies have explored AA’s wide variety of inflammation-related activities in different parts of the human body.
In a study related to the human kidney, for example, researchers with the International Journal of Molecular Sciences explored AA’s role in inflammatory damage and repair. They found that prostaglandins (PG), thromboxane (Tx), leukotrienes (LTs) and hydroxyeicosatetraenoic acids (HETEs) are the major metabolites generated from AA. Increased levels of prostaglandins in particular resulted in inflammatory damage to the kidney. As a result, they suggested that drug candidates continue to target the AA metabolism pathway in order to treat kidney inflammation 7.
Depression
In a study of twenty moderately to severely depressed patients, published by Lipids, researchers investigated the relationship between the severity of depression and ratios of n-3 and n-6 long-chain polyunsaturated fatty acids (PUFA) in plasma and erythrocyte phospholipids (PL). Findings indicated a significant correlation between the ratio of Arachidonic Acid to Eicosapentaenoic Acid (EPA) and severity of depression as rated by the HRS (Hamilton Rating Scale; P<0.05) and the LRS (Linear Rating Scale) for depression (P< 0.01). Researchers concluded that their findings provide a basis for studying the effect of nutritional supplementation that aims at reducing the AA/EPA ratio in depressed patients 8.
Hair Growth
In a study published by Annals of Dermatology, researchers examined the effect of AA on hair growth via in vivo and in vitro models. Specifically, the impact of AA on human dermal papilla cells (hDPCs) and hair shaft elongation was evaluated via MTT assay and hair follicle organ culture. The expression of cell growth and survival factors in hDPCs were then subsequently investigated with the Western blot test or via immunohistochemistry. AA was found to enhance the cell viability of hDPCs and promote the expression of several factors associated with hair growth, including fibroblast growth factor-7 (FGF-7) and FGF-10. Further, AA significantly promoted hair shaft elongation and increased the proliferation of cellular keratinocytes. Finally, AA was found to promote hair growth by prolongation of the anagen phase in telogen-stage laboratory mice 9.
Asthma
Leukotrines (LTs) and prostaglandins (PGs), two families of proinflammatory mediators arising in AA metabolism, have been implicated in the inflammatory activity that occurs in asthmatic airways. Given AA’s significant role in the inflammatory processes underlying asthma, new pharmacologic agents (such as the sulfidopeptide-LT antagonists, zafirlukast, montelukast, and pranlukast and the 5-lipoxygenase [5-LO] inhibitor, zileuton) have been developed with the goal of targeting specific elements of the AA metabolic pathway in the inflammatory cascade that occurs in asthmatic reactions. Further, numerous studies have indicated an imbalance of AA metabolism in platelets from asthmatic patients 10.
In a study published by Frontiers in Immunology, researchers reviewed the role of AA in the treatment and resolution of chronic lung inflammation in asthmatic patients. As has been discussed, AA is metabolized by two classes of enzymes, cyclooxygenases (COX), which produce prostaglandins (PGs), thromboxanes, and lypoxygenases (LOX), which form leukotrienes and lipoxins (LXs). In asthma, two primary pro-inflammatory mediators from COXs are PGE2 and the prostaglandin15-Deoxy-Delta-12,14-PGJ2 (15d-PGJ2). The LOX pathway produces LXA4 and LXB4. In the different models of asthma studied in this scientific review, PGE2, 15d-PGJ2, and the LX derivatives reduced lung inflammation and remodeling.
Further, they inhibited chemotaxis and the movement of several inflammatory cells implicated in asthma pathogenesis, such as eosinophils. However, while PGE2 and LXA4 demonstrated distinctly beneficial results in asthmatic patients, researchers did identify limitations to their clinical use, as PGE2 caused side effects and LXA4 presented low stability. Thus, they concluded that, though AA-derived SPMs did indeed induce resolution of both inflammatory response and tissue remodeling in asthma, safer and more stable analogs should be investigated further11.
Dosage
AA supplements are not approved by the FDA for any condition. As a result, there is no official recommended dose. Users and supplement manufacturers recommend dosages based primarily on trial and error, and thus consultations regarding supplementation should be conducted with patients’ physicians. AA supplementation is not recommended for individuals with chronic inflammatory disorders, such as rheumatoid arthritis or inflammatory bowel disease – instead, the ideal dose of AA may be quite low and indicate a dietary restriction thereof 1.
Side Effects
Arachidonic Acid (AA) supplementation is not reputed to cause significant side effects in clinical trial participants. With that being said, it is suspected that improper use of AA has the potential to cause negative immunological and cardiovascular effects, as reckless supplementation would upset the natural balance of omega-6 and omega-3 fatty acids in the human body. More long-term studies on humans are necessary to accurately assess the safety of AA supplementation.
References:
[1] El Ridi R. & Tallima H. (2017). Arachidonic acid: Physiological roles and potential health benefits – A review. Journal of Advanced Research, 11: 33–41.
[2] De Souza E.O., Fox C.D., Healy J.C., Joy J.M., Lopez H.L., Ormes J.A., et al. (2016). Effects of Arachidonic Acid supplementation on acute anabolic signaling and chronic functional performance and body composition adaptations. PloS one, 11(5): e0155153.
[3] Aasen K., Cameron-Smith D., Chan A., D’Souza R.F., Egner I.M., Figueiredo V.C., et al. (2018) Effect of dietary Arachidonic Acid supplementation on acute muscle adaptive responses to resistance exercise in trained men: a randomized controlled trial. Journal of Applied Physiology, 124: 1080-1091.
[4] Chen J., Wang B. & L. Wu (2021) Metabolism pathways of arachidonic acids: mechanisms and potential therapeutic targets. Signal Transduction and Targeted Therapy, 94(6).
5 Campbell B., Cooke M., Greenwood M., Harvey T., Iosia M., Jitomir J., et al. (2007) Effects of arachidonic acid supplementation on training adaptations in resistance-trained males. Journal of the International Society of Sports Nutrition, 4(21).
6 Aasen K., Cameron-Smith D., Chan A., D’Souza R.F., Egner I.M., Figueiredo V.C., et al. (2018) Effect of dietary Arachidonic Acid supplementation on acute muscle adaptive responses to resistance exercise in trained men: a randomized controlled trial. Journal of Applied Physiology, 124: 1080-1091.
7 Chen Q., Fu X., Gai Z., Patra J.K., Wang D., Wang T., et al. (2019) Arachidonic Acid metabolism and kidney inflammation. International Journal of Molecular Sciences, 20(15): e3683.
8 Adams P.B., Lawson S., Sanigorski A. & A.J. Sinclair (1996) Arachidonic Acid to eicosapentaenoic acid ratio in blood correlates positively with clinical symptoms of depression. Lipids, 31: S157-S161.
9 Cho A.R., Choi S.J., Eun H.C., Hang S., Kim K.H., Kwon O., et al. (2016) Role of Arachidonic Acid in promoting hair growth. Annals of Dermatology, 28(1): 55-64.
10 Yen S.S. & H.G. Morris. An imbalance of arachidonic acid metabolism in asthma. Biochemical and Biophysical Research Communications, 103(2): 774-779.
11 Carvalho V.F., Coutinho D.S., Ferrero M.R., & Insuela D.B.R. (2020) Could Arachidonic Acid-derived pro-resolving mediators be a new therapeutic strategy for asthma therapy? Frontiers in Immunology, 11: 580-589.