What is the definition of NAD?
NAD refers to nicotinamide adenine dinucleotide, a coenzyme that can found in all eukaryotic cells. NAD can also be defined as a pyridine nucleotide, which functions as a highly important cofactor in the soluble phase of essential reduction-oxidation processes in all living cells. A cofactor can be defined as an agent or substance, such as a coenzyme or an ion, which is fundamental to the functionality and stability of any given enzyme.
NAD is critical in the execution of numerous life-supporting cellular reactions. It catalyzes reduction-oxidation (or redox) reactions, or cellular activities that transfer electrons between chemical species, examples of which include photosynthesis, corrosion, combustion, and respiration. These reactions are all contingent on a cell’s ability to accept and transfer electrons. NAD in its oxidized form, NAD+, and in its reduced form, NADH, provides the cell with this mechanism.
Research has indicated that increased production of NAD(H/+) in the human body may yield numerous beneficial health effects and possibly elevate both health and lifespan by combating age-related neurodegenerative and physiological disorders. Because of the coenzyme’s life-supporting activities, it is popularly taken in a supplement form to combat hypercholesterolemia, hypertension, chronic fatigue syndrome, fibromyalgia, negative effects of alcohol on the liver and kidneys, and various age-related ailments and inconveniences, from hair loss to Alzheimer’s dementia.
What does NAD Do?
NAD is a critically important actor in the lives of cells. Life-and-death decisions made on a molecular level hinge on an optimal balance of NAD(+/H) and its ability to transfer electrons in redox processes. Several different types of dehydrogenases, which are enzymes that catalyze cellular respiration and essential metabolic reactions, must utilize NAD(+/H) in a variety of activities, from the breakdown of sugars and fats via glycolysis to the tricarboxylic acid cycle. NAD has also been revealed to be a key regulator of sirtuins, though this finding is relatively recent and its exact mechanisms of action are still being elucidated. However, increased sirtuin activity has been revealed in clinical research to extend the lifespan of organisms from yeast to small rodents. NAD may also inhibit the production of PARP1, an enzyme essential to DNA repair and transcription, which is considered a possibly effective treatment in the desensitization of tumour cells to chemotherapy-induced stress. Essentially, organisms require NAD for metabolic regulation, efficient synthesis of fats, proteins, and lipids, resistance to oxidative stress, DNA repair, endocrine signaling, in addition to a variety of other essential activities that are still being discovered.
NAD+ vs. NADH: What’s the difference?
In short, NAD+ is nicotinamide adenine dinucleotide in its oxidized form, while NADH is the coenzyme in its reduced form. Though they emerge from the same coenzyme, they have two opposing mechanisms, both of which involve catabolic electron transfer processes. Catabolism refers to the metabolic process that reduces molecules into smaller units that can then facilitate energy release. Anabolism, on the other hand, constructs molecules from smaller, reduced units.
NAD+ has a positive charge, and thus has the capacity to oxidize the biological material it interacts with. It is an electron acceptor, thereby reducing electrons from other molecules. Conversely, instead of oxidizing other biological compounds, NADH becomes oxidized through metabolic processes. When NADH reduces another compound, it becomes oxidized into NAD+. These alternating redox processes are essential in the support of enzymatic activity; in other words, without coenzymes, most enzymes are far less useful. Without NAD, many eukaryotic cells cannot carry out their life-supporting processes at all.
NAD+ and NADH have both emerged in clinical research as integral in regulating metabolic function, energy release, and stress resistance.
NAD+ and NADH Benefits and Uses
Anti-Aging (NAD+)
NAD+ has been touted by some researchers as the “Molecule of Youth” thanks to its clinically validated anti-aging properties. There have been numerous studies verifying nicotinamide adenine dinucleotide’s role in ameliorating age-related physical and neurodegenerative diseases and even inhibiting the physiological signs of aging. A 2014 study published by Trends in Cell Biology revealed that a natural decline in NAD+ may be directly linked to a decrease in sirtuin (proteins located in mitochondria that are associated with aging, cellular protection and regulation, and metabolism) activities. It follows that countering this decline with NAD+ supplementation may ameliorate age-associated pathophysiologies associated with altered sirtuin activities. A 2013 study published by Cell Press had similar findings; researchers found that a decline in nuclear NAD+ was partially responsible for an alternate PGC-1α (which is a key regulator of energy metabolism)/β-independent pathway of nuclear-mitochondrial interface, which resulted in age-related metabolic and energy decline. They then discovered that increasing NAD+ levels in elderly mice reversed this process and restored “youthful” mitochondrial function. Further, recent research has confirmed that consistent nicotinamide riboside supplementation (with the objective of elevating NAD+ levels specifically) is well tolerated over long periods of time in healthy older adults.
Skin Care (NAD+)
NAD+ and its youth-restoring benefits may possibly extend to the skin, though some of its mechanisms related to skin protection and regeneration are not well understood. Research has revealed that niacin deficiency – and by extension, low NAD+ levels – can induce increased sun sensitivity in the skin. Further, researchers at the College of Pharmacy and Arizona Cancer Center at the University of Arizona found that NAD+-dependent cellular signaling activities may be critical in identifying skin carcinogenesis early on. These and other data suggest that elevated NAD+ levels may be influential as potential skin cancer prevention agents.
From a cosmetic perspective, NAD+ has been hypothesized to have an anti-aging effect on the skin as a result of its impact on sirtuin activities. Sirtuin stimulation has been validated in clinical trials to have antioxidant properties as well as protective properties against photodamage to the skin. However, the functional relationship between NAD+ and mitochondrial sirtuins must be better understood before NAD+ can be specifically validated as a skin care agent.
Addiction (NAD+)
In recent years, some attention has been dedicated to NAD+ and its potential ability to combat drug and alcohol addiction. This idea emerges from numerous studies implicating low levels of NAD+ in vulnerability to addiction. Proponents of intravenous NAD+ treatment cite a 70-80% success rate in its ability to assuage cravings and withdrawal in individuals addicted to prescription drugs, opiates, benzodiazepines, stimulants, cocaine, suboxone, methadone, and alcohol. Several addiction centers that use NAD+ IV therapy as a mainstay in their rehabilitative programming have been erected over the past decade. However, clinical studies regarding NAD+ and its efficacy in combating addiction are very limited, reportedly due to a lack of financing.
Hair Loss (NAD+)
Research validating NAD+ and its role in preventing alopecia, or patterned hair loss, is limited. However, some findings have indicated that the coenzyme has potential in this arena. For example, a 2013 study conducted across several universities in the United States, Japan, and China found that NAD+ suppresses melfloquine-induced oxidative stress (with melfloquine referring to an antimalarial pharmaceutical) that causes hair cells to degenerate. This research plus some existing anecdotal evidence recommends more research into the relationship between NAD+ and hair growth and regeneration.
Grey Hair (NAD+)
To date there have not been any clinical studies specifically addressing NAD+ and its ability to combat age-related hair loss/alopecia and/or graying hair. However, some clinical and anecdotal evidence indicates that there may be curative potential in this arena. Given nicotinamide adenine dinucleotide’s overall anti-aging activity and control over numerous aspects of gene expression, it follows that NAD+ may have an anti-aging effect that extends to age-related hair loss and coloring. Public platforms from Harper’s Bazaar to Reader’s Digest have discussed the anti-aging potential of NAD+ for both health and cosmetic purposes, but further research is necessary in the arena of age-induced alopecia and graying.
Metabolism (NAD+)
Nicotinamide adenine dinucleotide plays a critical role in metabolic functioning. As such, NAD+ may be essential in treating various metabolic diseases. For example, derailed NAD+ homeostasis is known to contribute to the development of type 2 diabetes and fatty liver disease.
Research has also found that a 10 to 40% decrease in caloric intake (known as healthy caloric restriction) has been known to improve metabolic function by stimulating sirtuins to sense NAD+. Nicotinamide riboside, which increases NAD+ concentrations, has also been observed to increase oxidative metabolism and protection against metabolic abnormalities induced by a high-fat diet through activating sirtuins (specifically, the groups classified as SIRT1 and SIRT3).
Kidney Function (NAD+)
In a 2014 study published by Kidney International, researchers addressed NAD+ and its role as a therapeutic agent against chemotherapy-induced kidney dysfunction. Results of this study indicated that, by regulating NAD+ levels via NQO1 (a metabolic and cellular protective protein) activation, researchers were able to significantly decrease inflammatory cytokines and markers for acute renal damage in laboratory mice treated with the chemotherapeutic cisplatin.
There have also been numerous studies exploring NAD+ and its various activities as a coenzyme insofar as they regulate prostaglandins in the kidneys, with prostaglandins being hormones that play key roles in indicating and modulating tissue damage and inflammation. For example, a 2006 study conducted between National Cheng-Kung University in Taiwan and the University of Kentucky in Lexington, Kentucky found that a NAD+-dependent 15-hydroxyprostaglandin dehydrogenase (15-OH-PGDH), which was isolated from porcine kidneys, was able to ameliorate renal oxidative stress by deactivating particular chemical enzymes.
Hypertension (NADH/NAD+)
In a study conducted at Georgetown University Medical Center in Washington, D.C., researchers addressed NADH and its ability to prevent or ameliorate elevated blood pressure levels associated with aging. They found that continuous oral NADH administration observed lower total cholesterol and systolic blood pressure in spontaneously hypertensive rats.
Further, a 2018 study conducted at the University of Colorado Boulder explored NR’s (or nicotinamide riboside, a natural stimulating agent for the production of NAD+) role in modulating blood pressure and arterial health. Researchers administered 500 mg of NR chloride twice daily to twelve healthy men and women aged 55 to 79 for six weeks, while the control group received NR for the first six weeks, followed by placebo. Results yielded that 1,000 mg of NR daily elevated NAD+ levels by approximately 60%. Further, in 13 participants with stage 1 hypertension, systolic blood pressure dropped significantly after NR supplementation.
Chronic Fatigue (NADH)
In a 1999 study published by the Annals of Allergy, Asthma & Immunology, researchers evaluated the efficacy of orally-administered NADH in the alleviation of symptoms associated with Chronic Fatigue Syndrome (CFS). The results of the study indicated that 31% of supplemented individuals responded positively to NADH in contrast to the placebo group, which responded at an 8% positivity rate. Researchers concluded that further studies of NADH and its ability to ameliorate neurocognitive dysfunction and other ailments associated with CFS were advisable. Since this study was conducted there have been numerous follow-up trials over the years that have explored NADH’s efficacy in alleviating CFS-related symptoms. This is significant, as there is no known cure for the chronic illness.
A 2010 study examined the efficacy of NADH supplementation in patients with CFS for three months. After conducting a stress test, researchers observed that an oral 20 mg dose of NADH was associated with a decrease in anxiety with supplemented patients. However, NADH supplementation alone did not yield very significant changes in the variables explored with afflicted patients, including fatigue, functional performance, sleep quality, and exercise capacity.
In 2014, researchers from several universities based in Barcelona, Spain found through an eight-week placebo-controlled trial that supplementation of oral CoQ10 (200 mg) plus NADH (20 mg) daily had significant therapeutic benefits for subjects with CFS. Specifically, supplemented individuals showed a significant reduction on the fatigue impact scale (p<0.05). Researchers also reported higher citrate synthase and lower lipoperoxides in blood mononuclear cells of the supplemented group.
Fibromyalgia (NADH)
Chronic fatigue syndrome is often, though not always, co-morbid with fibromyalgia, a syndrome that induces chronic and unexplainable pain in the muscles and soft tissues. Most existing research addresses NADH and its role in alleviating ailments related to CFS principally, but given the overlap in symptoms (musculoskeletal pain and fatigue, sleep disturbances, and cognitive dysfunction), it can be inferred that NADH may be effective in the treatment of fibromyalgia as well. For example, the International Journal of Tryptophan Research recommends NADH supplementation to counter the metabolic dysregulation inherent in both Chronic Fatigue Syndrome and fibromyalgia.
Depression (NADH)
NADH has gained some repute as a potential antidepressant and mild anxiolytic. Some users have purported that NADH supplementation has produced a mental energizing effect, thus combating the malaise associated with clinical depression. The notion of NADH as an antidepressant began to gain traction as early as 1991, when researchers J.G.D. Birkmayer and W. Birkmayer conducted an open-label trial of NADH’s efficacy in treating 205 patients suffering from clinical depression. Results revealed that 93% of patients experienced amelioration of depressive symptoms. However, this type of trial, which does not use placebo, is notorious for creating bias in both the researcher and the subjects of the trial and is overall considered to be weak in design. Therefore, this study should not be taken as definitive.
The only known placebo-controlled, laboratory-design trial assessing NADH and its efficacy as an antidepressant was conducted in 2004 and published by Pharmacology, Biochemistry and Behavior. In this study, researchers from the Institute of Pharmacology and Toxicology in Berlin, Germany evaluated the impact of NADH supplementation on rats with depressive symptoms. Results indicated that, at a minimum effective dose of 5 mg, NADH demonstrated an antidepressant-like effect in laboratory Wistar rats subjected to the forced swim test. No human trials have been conducted to date.
To conclude, NADH may have antidepressant potential, but much more research is required to confirm this capability.
Weight Management (NADH)
In a study conducted across various universities across South Korea, researchers explored the pharmacological effects of NADH oxidation in obese mice. Results indicated that NADH provoked mitochondrial fatty acid oxidation, consequently attenuating glucose intolerance, fatty liver, dyslipidemia, and increased adiposity in mice (all factors contributing to and/or resulting from obesity). Further, treated mice also demonstrated higher expressions of genes related to mitochondrial energy metabolism.
Some clinical evidence also indicates that the effective balancing of NAD+ and NADH may be a potentially effective antidiabetic strategy. A study conducted across U.S. and Chinese medical universities found that a consequence of NADH/NAD+ redox imbalance is oxidative stress on lipids and proteins, eventually leading to the development of diabetes and associated complications, such as insulin resistance and obesity.
Parkinson’s (NADH)
While research into NADH’s efficacy in treating Parkinson’s disease is limited, some researchers have hypothesized that its mechanism of stimulating tyrosine hydroxylase and dopamine biosynthesis may indicate a potential to ameliorate Parkinson’s symptoms, such as tremors and impaired balance. Further, a study conducted at St. Josef-Hospital at Ruhr-University of Bochum in Germany found that NADH administration significantly increased the biological synthesis of plasma levodopa, a primary medication used in the treatment of Parkinson’s disease. However, further placebo-controlled trials are necessary in order to validate any clear benefit from NADH on its own in the treatment of Parkinson’s disease.
Liver (NADH)
Nicotinamide adenine dinucleotide’s role in fatty acid oxidation and/or synthesis in the liver is fairly well-understood. Elevated levels of glycerol 3-phosphate after alcohol consumption are linked to an imbalance in NAD(H/+), resulting in the direct esterification of fatty acids and the development of alcoholic fatty liver, a dangerous condition that can lead to steatohepatitis, fibrosis, and/or cirrhosis. Recent research has been investigating NADH and its therapeutic potential in reversing fatty liver disease, both in relationship to alcohol abuse and the natural aging process of the liver. A 2016 study conducted by Second Military Medical University in Shanghai, China found that elderly mice with fatty liver disease displayed decreased oxidative stress, hepatic inflammation, and insulin sensitivity when administered oral nicotinamide riboside, a natural NAD precursor. These data and other similar research recommend further investigation into NAD+ and its therapeutic potential for hepatic diseases.
How to increase NAD?
NAD+ and NADH are both readily available for direct ingestion via oral supplements, though isolation and synthesis of these molecules are relatively new processes. Supplements that are natural precursors to NAD+ and/or NADH, most notably nicotinamide riboside, are also commercially available as nutritional supplements. Nicotinamide riboside is arguably the most efficient way to use supplementation to achieve an optimal balance of NAD+/NADH in the human body, and is linked to the sustainable prevention of hepatic diseases, metabolic syndrome, diabetic symptoms, and age-related neurodegenerative diseases by facilitating the rejuvenation of “youthful” NAD.
Other ways to increase NAD and activate its associated benefits include caloric restriction (limiting intake by 10%-40% to avoid malnutrition) and regular exercise. Food sources that provide NAD+ and/or NADH include milk, fish, poultry, yeast, leafy green vegetables, and some types of mushrooms, including the Crimini mushroom.
Examples of NAD+-Dependent Enzymes
Examples of enzymes that depend on NAD+ to catalyze life-supporting oxidation-reduction reactions include:
Aldehyde Dehydrogenase
Aldehyde dehydrogenase oxidizes aldehydes, compounds that are found in many essential oils, into organic carboxylic acids. This enzyme requires NAD+ to catalyze a chemical reaction that results in three products: acid ions, NADH, and a hydrogen ion. Aldehyde dehydrogenase participates in a total of seventeen metabolic pathways, among which include glycolysis, ascorbate/aldarate metabolism, bilic acid biosynthesis, Urea Cycle, metabolism of amino acids, tryptophan metabolism, pyruvate metabolism, and fatty acid metabolism.
Lactate Dehydrogenase
Lactate dehydrogenase is essential to anaerobic glycolysis, which refers to the process by which glucose is converted to lactate in circumstances where limited oxygen is available. This type of glycolysis is primarily functional during periods of brief but intense exercise, and provides short-term bursts of energy to an organism. An essential element of this process involves the conversion of pyruvate to lactate in conjunction with the conversion of NADH to NAD+.
Pyruvate Dehydrogenase
Pyruvate dehydrogenase deficiency results in some of the most common disorders in pyruvate metabolism, the most common symptom of which is a potentially fatal buildup of lactic acid. The regulation of pyruvate dehydrogenase requires three primary coenzymes: thiamine pyrophosophate, lipoamide, and flavin adenine dinucleotide, the latter of which transfers two hydrogen atoms to lipoamide via the substrate NAD+.
Alcohol Dehydrogenase
Alcohol dehydrogenase oxidizes alcohol into aldehydes and/or ketones. This enzyme uses a zinc atom alongside a large NAD+ coenzyme to essentially detoxify alcohol, making it the human body’s primary defense against ethanol. Alcohol dehydrogenase is able to quickly convert acetaldehyde, a toxic substance, into acetate, which can be easily consumed by cells. However, this enzyme can also use NAD+ to alter other alcohols, such as methanol, into dangerous and undrinkable substances by converting them into formaldehyde, which is fatally toxic.
NAD+ and NADH Dosage
A safety assessment of nicotinamide riboside, a precursor to NAD+, conducted in 2016 confirmed that no adverse effects were observed at a maximum dose of 300 mg/kg daily. An earlier safety assessment published by the Journal of Environmental Pathology, Toxicology, and Oncology found that the maximum safe intravenous dose of NADH tolerated by Beagle dogs was 500 mg/kg daily.
However, physicians recommend a reduced dose of NR/NAD(+/H) in order to avoid any unpleasant consequences from an induced NAD+/NADH imbalance. For nutritional purposes and energy enhancement, a daily dose of 2.5-5 mg orally is advisable. For individuals suffering from Chronic Fatigue Syndrome, some physicians recommend a daily dose of 10-15 mg. Supplementation is largely dependent on the health and metabolic makeup of the individual, so speak to a physician before beginning any sort of NAD or NR regimen.
NAD Therapy
NAD therapy typically refers to an intravenous form of NAD supplementation, which allows the molecule to bypass the digestive process and undergo rapid absorption. NAD IV therapy for the purposes of addiction and recovery is relatively new, and not enough research has been conducted in order to clinically verify its long-term effectiveness. However, proponents of repeated intravenous NAD administration cite a 70-80% efficacy rate in terms of alleviating substance cravings and facilitating detoxification without unpleasant withdrawal symptoms.
Intravenous NAD therapy has also gained some traction as an innovative “brain restoration” treatment, with alleged benefits including increased concentration, alertness, and mental clarity, improved memory, and elevated mood via the stimulation of dopamine, serotonin, and noradrenaline production. Typically, this type of therapy can take place over a series of four to fourteen days, depending on the condition of the individual seeking treatment.
NAD Structure and Biology
Nicotinamide adenine dinucleotide is essentially the vitamin niacin in its coenzyme form. Niacin, or vitamin B3, is one of eight essential B-complex, water-soluble vitamins that support the body in converting carbohydrates, proteins, fats, and sugars into usable energy. Niacin is also a component of nicotinamide adenine dinucleotide phosphate (NADP), which acts primarily on the metabolism of fats and sugars. While NAD+ refers to nicotinamide adenine dinucleotide in its oxidized form (with the + designation referring to the positive charge on its nitrogen atom), NADH refers to the molecule in its reduced form.
NAD’s importance lies primarily in its ability to catalyze mitochondria, which are the tiny intracellular “powerhouses” that are integral to cellular functioning and cellular energy metabolism. They, in turn, stimulate the production of the organic chemical adenosine triphosphate (ATP), often referred to as the “molecular unit of currency” in energy transfer processes. In order to function efficiently, eukaryotic cells require an optimal ratio of NAD+/NADH. If the mitochondrial powerhouses of these cells are not able to carry out essential life-supporting processes effectively, the result is mitochondrial dysfunction, which has been associated with over forty serious diseases including Alzheimer’s disease and type 2 diabetes. Some life-supporting interactions facilitated by mitochondria and ATP production are nitrogen metabolism via the Urea Cycle, fat and sugar breakdown and metabolism via ketone formation, and calcium storage, among many others.
Mitochondrial ATP production cannot be executed without NAD acting as a coenzyme. In this process, NAD acquires two electrons and a proton from enzymes during aerobic respiration (or the Krebs cycle). The result is NADH, the reduced form of nicotinamide adenine dinucleotide. The transition between NAD+ to NADH involves an exchange of hydrogen atoms and electrons between coenzymes and enzymes. Whereas NAD+ acts as an electron acceptor in catalyzed reactions, NADH is an electron donor. While undergoing oxidization, NADH donates electrons to Complex I, the large enzyme that catalyzes the electron transport chain process. This flow of electrons is critical to ATP biosynthesis and the health and maintenance of cells.
Some of the many enzymes that require NAD as a coenzyme include alcohol dehydrogenase, lactate dehydrogenase, glyceraldehyde phosphate dehydrogenase, malate dehydrogenase, and pyruvate dehydrogenase.
The molecular formula of NAD+ is C21H26N7O14P2. The molecular formula of NADH is C21H29N7O14P2.
NAD Molecule
The molecule NAD, in both its oxidized and reduced forms, serves as the eukaryotic cell’s primary hydrogen carrier in the execution of essential oxidation-reduction (redox) reactions. It has also emerged in recent research as an important “signaling molecule,”
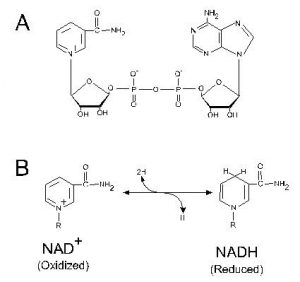
which triggers cellular mitochondria and sirtuins, a class of proteins that heavily influence aging and age-related disease, to begin various integral developmental processes on a molecular level.
NAD has been described by many researchers as the “Golden Nucleotide” and the “Molecule of Youth.” Research has yielded that the restoration of NAD levels in elderly or diseased humans and mammals promotes cardiovascular and renal health and may extend the lifespan overall. As such, researchers are in continuous pursuit of verifiably safe and efficient NAD-boosting treatment options that may better protect the body against age and disease.
NADH Mechanism
Metabolic processes can be broken down into two primary parts: anabolism and catabolism. Whereas an anabolic pathway involves the endogernic use of energy (in other words, energy is absorbed from a cell’s environment), catabolic pathways involve the exergonic explusion of energy, in which large molecules are broken down into smaller units. In catabolic cellular reactions (for example, glycosis, or the breaking down of glucose molecules to produce ATP), NADH is essential, with NAD+ serving as an oxidizing agent.
NADH and Cellular Respiration
NADH plays a crucial role in cellular energy production and cellular respiration. Cellular respiration can be defined as a series of metabolic reactions that convert energy synthesized from nutrients into adenosine triphosphate (ATP) and subsequently release excess waste, carbon dioxide, and water. These processes are typically completed over a series of four steps: glycosis (which involves the metabolic breakdown of sugars intopyruvate), Acetyl-CoA formation (a conversion process that transforms pyruvate into Acetyl-CoA, a molecule that modulates protein, carbohydrate, and lipid metabolism), Krebs cycle (which involves the oxidation of Acetyl-CoA and the subsequent release of electrons), and Electron Transport Chain (or ETC, which converts high energy molecules, such as NADH, into low energy acceptor molecules, such as oxygen). It is estimated that, during cellular respiration, each NADH molecule produces three ATP molecules.
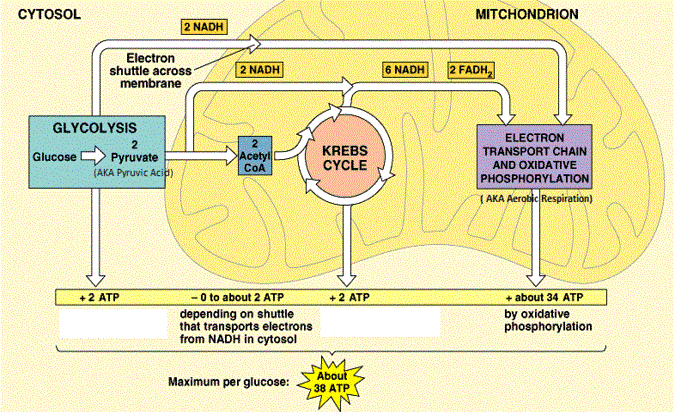
If cellular respiration is not completed correctly, organisms can develop mitochondrial oxidative phosphorylation disorders, which can manifest in seizures, muscle weakness and/or hypotonia, cardiomyopathy, and/or ophthalmoplegia. Mitochondrial mutations resulting from oxidative stress and inefficient cellular respiration have also been linked to diabetes, Parkinson’s disease, Alzheimer’s disease, and even deafness.
NAD Levels
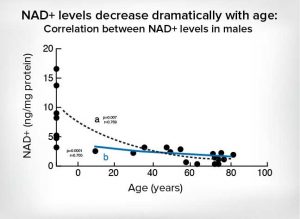
Nicotinamide adenine dinucleotide is a life-supporting coenzyme and a signaling molecule, or an agent that triggers developmental processes on a cellular level. As vertebrate organisms age, our NAD levels inevitably decline. Low NAD levels are associated with cellular oxidative stress, which can contribute to metabolic syndrome, fatigue, cardiovascular diseases, multiple sclerosis, thyroid dysfunction, and inflammation. By the time an individual reaches middle age, levels of NAD have naturally reduced to roughly half of their youthful quantity.
Several studies conducted over the past decade have confirmed that increasing NAD levels can yield notable positive effects, from direct reversal of chronic mitochondrial dysfunction to an overall extension of lifespan. While the exact mechanisms that cause NAD levels to decrease with age are still being studied and elucidated, a 2016 study published by Cell Metabolism made a significant breakthrough in implicating CD38, a glycoprotein, which appears to increase with age and decompose nicotinamide adenine dinucleotide in vivo.
NAD Side Effects, Safety, Dangers and Warnings
Doses exceeding 5 mg per day of NAD or its precursors for extended periods have been demonstrated to cause insomnia, agitation and/or anxiety, and gastrointestinal issues. For this reason, nicotinamide adenine dinucleotide supplementation should not continue beyond a twelve-week period.
Clinical studies assessing the safety of nicotinamide adenine dinucleotide or nicotinamide riboside for children, pregnant or nursing women, or individuals with severe liver or kidney abnormalities have not been conducted to date. Therefore NAD(+/H), or NR supplementation is inadvisable for these groups.
At present, no contraindications between NAD+/NADH/NR supplements and any other medications are known.
References:
https://www.sciencedirect.com/science/article/pii/S0014256510001591?via%3Dihub
https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1473-2165.2008.00354.x
https://www.ncbi.nlm.nih.gov/pubmed/19149600
https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1473-2165.2008.00354.x
https://www.ncbi.nlm.nih.gov/pubmed/15312041
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3680915/
http://journals.sagepub.com/doi/abs/10.4137/IJTR.S11193
https://www.ncbi.nlm.nih.gov/pubmed/9893217
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3616234/
https://www.ncbi.nlm.nih.gov/pubmed/26791540
http://journals.plos.org/plosone/article/file?id=10.1371/journal.pone.0079817&type=printable
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5686634/
https://www.sciencedirect.com/science/article/pii/S1081120610625951
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2871241/
https://www.ncbi.nlm.nih.gov/pubmed/18419308
https://www.sciencedirect.com/science/article/pii/S0092867413015213
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5508210/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4112140/
https://www.sciencedirect.com/science/article/pii/030441659090115D
https://www.liebertpub.com/doi/full/10.1089/ars.2014.6181
https://www.sciencedirect.com/science/article/pii/S0085253815562212
https://www.sciencedirect.com/science/article/pii/B9780323034104500122
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2633431/
https://www.ncbi.nlm.nih.gov/pubmed/14751458
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5573868/
https://www.sciencedirect.com/science/article/pii/S1550413116302443
https://pubchem.ncbi.nlm.nih.gov/compound/nadh#section=Top
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3683958/
https://www.ncbi.nlm.nih.gov/pubmed/9805207
http://science.sciencemag.org/content/352/6282/231
http://www.biochemist.org/bio/03701/0009/037010009.pdf
http://science.sciencemag.org/content/350/6265/1208
https://www.sciencedirect.com/science/article/pii/B9780123809209000031




that was the most thorough report of nad/nadh I’ve read (and was able to understand). Thanks
Hy K.